A Tale of Two Scientists
How Thomas Midgley Jr. poisoned the world—and Clair Patterson saved it.
I. The Man Who Measured the Age of the Earth
Clair Cameron Patterson was born on June 2, 1922, in a small town in central Iowa called Mitchellville. His father was a mail carrier. His mother served on the local school board. When he was very small, he asked his mother: “Mommy, why is a drop of water round?” She responded by buying him a chemistry set, igniting his lifelong love of science. And it’s a good thing she did — a good thing for all of us. Clair Patterson’s love of science would, one day, quite literally save the world.
By eighth grade, he’d built a laboratory in his basement: a bench, shelves, beakers, Bunsen burners. When his uncle gave him a college chemistry workbook in ninth grade, the young Clair Patterson devoured it. Clair Patterson taught himself chemistry. He taught his high school teachers chemistry, too. “The teacher would say atrocious things that were totally wrong. And then I would get up and give a little explanation of how it really was,” Patterson would reminisce with a chuckle, years later, in a 1995 interview with journalist Shirley K. Cohen. “The science teacher would say something about electricity being a fluid, and I had to explain to them about electrons.”
He graduated high school at the age of 16 and enrolled at Grinnell College, a private liberal arts school in Grinnell, Iowa, about fifty miles east of his hometown. “I was the renegade,” Patterson recalled of his college years. He drank. He smoked. He didn’t do his homework. He disregarded the lab exercises his professors assigned him, and conducted experiments of his own instead. “In my third year, I blew up one end of the organic lab doing some experiments,” Patterson told Cohen. “You see, I would go beyond what I was supposed to be doing. And I was doing something with some diazo compounds. They were very fragile and unstable things. And I wanted to do some molecular chemistry and that sort of junk. Well, it blew up[!] . . . Oh, I loved physical chemistry, because I could do all sorts of things.”
Fittingly, it was Clair Patterson’s love of chemistry that led him to the other love of his life: a young woman named Laurie McCleary. They studied chemistry together at Grinnell. They started dating, fell in love, “became bonded,” in Patterson’s words. They married a few years later, in 1944, after they’d both earned master’s degrees at the University of Iowa.
They joined the Manhattan Project as civilians that same year, working first at the University of Chicago and then at the Clinton Engineer Works facility in Oak Ridge, Tennessee, where they lived in a “little military-operation-type” house in the hills, commuting by bus each day with their small dog. At Oak Ridge, Patterson specialized in mass spectrometry, a lab technique that, in the simplest possible terms, identifies what things are made of on the molecular and atomic levels. Patterson used the then cutting edge technology to characterize and analyze isotopes of uranium for the development of the atomic bomb. It was work that would prove instrumental to his later career, but he would come to regret working on such a “hideous weapon of warfare.” His superiors, Patterson reflected, “conveyed to young people like me that this is the thing to do. This hideous crime we were committing was — it was necessary.” He didn’t think of the Manhattan Project as a “crime” at the time, but that’s how he described it, half a century later.



After the war, the Pattersons went back to Chicago. Laurie, a trained scientist in her own right, found work as an infrared spectroscopist, supporting Clair while he pursued his doctorate at the University of Chicago. His dissertation was ambitious: he would determine the age of the Earth itself.
The method with which he would do this was elegant in theory. It had to do with zircon crystals. Zircon crystals are tiny, super-durable mineral crystals that form in igneous rocks and can endure billions of years of geologic upheaval; they can be found in some of our planet’s oldest rocks. As these crystals form, they trap uranium atoms, but they reject atoms of lead. As uranium decays over billions of years, however — uranium-238 has a half-life of 4.468 billion years — it transforms into lead. By measuring the ratios of uranium to lead in these ancient minerals using mass spectrometry, Patterson could calculate how long this decay process had been occurring, and thus the age of the rocks, and of the planet. “It’ll be duck soup, Patterson,” Harrison Brown, Patterson’s mentor and thesis advisor, told him.
There was a problem, however. Patterson’s measurements kept coming out wrong. His samples contained too much lead — far more than could have come from uranium decay alone. His entire laboratory, himself and his zircon crystals included, was contaminated with lead. “There was lead coming from here, there was lead coming from there; there was lead in everything that I was using,” Patterson explained to Cohen. “It was contamination of every conceivable source that people had never thought about before.” The contamination was everywhere: in the air, on his hands, in his hair. At first — for years, in fact — this was merely a frustrating obstacle to his research. He didn’t yet understand it was a symptom of a global catastrophe.

Patterson earned his PhD in 1951. He hadn’t yet solved that lead contamination problem, or measured the age of the Earth. (He’d written his dissertation on the uranium-lead dating method he’d pioneered instead, though he hadn’t yet succeeded in using it to determine how old the Earth was.) He wrote a proposal to the Atomic Energy Commission asking for postdoctoral funding to continue this work. They turned him down. They weren’t interested in knowing the age of the Earth, they told him. How was that information going to benefit the AEC? Patterson was devastated. “I cried on Harrison’s shoulder,” he told Cohen. Harrison Brown, who was then famous for his own contributions to the Manhattan Project, rewrote the proposal in his name, emphasizing the research’s practical applications. (If the AEC knew how old the Earth was, they could better estimate the planet’s supply of uranium ore to be used in nuclear weapons and reactor fuel, for example.) “Boom!” said Patterson. “I was awarded a fellowship, a postdoc, and that’s the money I used to begin.”
Patterson followed Brown to the California Institute of Technology the following year, where the Atomic Energy Commission continued funding his work. Determined to solve that lead contamination problem — and thus, to calculate the age of the Earth — Patterson did something that no scientist had ever done before: he built the world’s very first ultra-clean laboratory from scratch. He sealed his lab against outside air, acid-washed every tool, purified his own chemicals, controlled temperature, pressure, and air flow with obsessive precision. His colleagues thought him eccentric. Some thought he was nuts. One later described him as “intense times ten to the third power.” (These practices, radical for their time, have since become the gold standard for laboratories of all kinds worldwide.)

He became fanatical about contamination. “You know Pigpen, in Charlie Brown’s comic strip, where stuff is coming out all over the place?” he asked Cohen. “That’s what people look like with respect to lead. Everyone. The lead from your hair, when you walk into a superclean laboratory like mine, will contaminate the whole damn laboratory. Just from your hair!” He is said to have wandered the Caltech campus collecting bird shit during this time, which he glued to the side of his mass spectrometer as a reminder to his assistants: “If crappy samples go in, crappy results come out.”

With his laboratory clean — “superclean,” in Patterson’s words — Patterson returned his attention to the Earth and its antiquity. There was a better way to measure the planet’s age than zircon crystals, he realized: meteorites! Meteorites, which are essentially just chunks of broken asteroids, have been around since the birth of our solar system, unchanged since that ancient moment when a cloud of interstellar gas and dust collapsed, which scientists think was probably triggered by a shockwave from a distant, exploding star. Meteorites are time capsules, in other words; their lead and uranium isotopes preserve a record of when the solar system — and the Earth — formed. Our planet and our solar system and our solar system’s meteorites all share the same birthday. If Clair Patterson could determine the age of a meteorite, then — any meteorite — he could determine the age of the Earth.

Patterson got his hands on a piece of the Canyon Diablo meteorite, which slammed into what is now Arizona about 50,000 years ago, creating the Barringer Crater. He carried out the finicky, contamination-prone chemical separation work in his “clean room” at Caltech, then flew back to the Argonne National Laboratory in Illinois to use the institution’s brand-new, state-of-the-art mass spectrometer to analyze the samples. He published his findings in a 1956 paper titled “Age of meteorites and the earth.” The Earth, Clair Patterson determined, is 4.55 billion years old, plus or minus 70 million. It was one of the most momentous measurements of time ever made, and it has remained essentially unchanged for seventy years. Patterson was thirty-four years old.
That’s where most researchers would have stopped. It’s the kind of finding a scientist can hang his hat on. Professorship. Tenure. Prestige. All were sure to follow, like the sequential steps of a cascading chemical reaction. “You’ll be famous,” Brown had told Patterson, a decade earlier, when Patterson was still a graduate student, “because you will have measured the age of the Earth!” Clair Patterson cared for none of it.
It was understanding that Clair Patterson was after. Why is a drop of water round? How old is the planet on which we live? Why was his lab so completely and totally contaminated with lead!? Why was everyone and everything contaminated with lead? Where was all this lead coming from? Was this natural, or was it manmade?
Decades later, Shirley Cohen would ask him: “What was your motivation at this point? Were you thinking in an environmental sense?”
“No, I was not!” Patterson replied. “Science, science, science! I wanted to know, ‘What is this natural level of lead?’ I didn’t care two hoots about verifying what the contamination was. I was forced to measure the contamination in order to arrive at what was the natural level.”
He began by analyzing ocean sediments from the floors of the Atlantic and Pacific. Lead occurs naturally in ocean sediments, in small amounts. Over millions of years, rocks — some of which contain lead — are weathered and eroded and washed by rain into streams and rivers and ultimately, the ocean, where these sediments — and the lead they contain — settle on the ocean floor. (This is how, bit by bit, ocean floors are composed.) Zooplankton, too, contribute a little bit of lead to ocean bottoms. When these microorganisms are alive, floating in the ocean, they collect soluble lead — lead that is dissolved in water — “on the outside of their little bodies,” as Patterson put it. When these microorganisms die, their little bodies — and their lead — sink to the seafloor and become part of the sediment. With these natural processes in mind, Patterson expected to find some lead on the floors of the Atlantic and Pacific. But the levels he found were off the charts — about one hundred times greater than these natural processes could explain. Something is wrong here, Patterson thought.
He sailed the Pacific — getting “sicker than a dog,” he said — and measured lead in the ocean’s waters at various depths. Water sampled from the surface of the Pacific contained about twenty times more lead than water taken from the deep ocean, Patterson found. Lead seemed to be falling from the sky, he realized, settling on the ocean’s surface with rain and snow and the pull of gravity.
He kept looking up. He climbed Lassen Peak in northern California and collected snow some seven thousand feet above the sea. The data confirmed his suspicions. Lead levels in the mountains were between ten and one hundred times greater than at sea level. Lead was airborne, and a lot of it.
He made some calculations. He took the data he’d gathered from the Atlantic and Pacific and extrapolated it to estimate the total amount of lead that had made its way into all the world’s oceans. “It could easily be accounted for by the amount of lead that was put into gasoline and burned and put in the atmosphere,” Patterson found. “We had more tons put in the atmosphere from lead gasoline than we could see in the upper part of the world’s oceans right there.” This discovery put Patterson in an uncomfortable position. Oil companies were funding his research at the time. We’re in serious trouble, Patterson thought.
But the boy who couldn’t keep quiet when his high school teachers said “atrocious things” couldn’t keep quiet about this, either. He published his findings in Nature in 1963. “I wrote a big paper,” Patterson told Cohen, “and I said ‘This lead is coming from leaded gasoline.’ Wham! They stopped my research.” Not only did the oil companies stop funding his research, they tried to get the Atomic Energy Commission to stop funding him, too. “They went around and tried to block all my funding,” Patterson said. Colleagues, including Harrison Brown, started distancing themselves from him. Prestigious universities invited him to come lecture about his pioneering work on the age of the Earth. He lectured on lead pollution instead. He wasn’t invited back. Oil industry reps materialized on his doorstep and tried to buy him out; he told them to get off his property. Clair Patterson was the renegade once again.

A renegade who was running out of funding, however. “I needed money,” he told Cohen, “a lot of money, because since I got this idea about lead coming from gasoline, I wanted to look at the record. Where do you see that record? You see it in the snow that never melts in the polar regions. It comes out of the air, which has lead in it. Lead is in snowflakes. It goes down, and you have a layer there. Next year you have another one.”
“Did you already have these [ice] cores?” Cohen asked him.
“No,” he told her. “No way whatsoever.”
This was another one of Clair Patterson’s radical ideas — an idea that revolutionized environmental science. Glaciers in the polar regions of our planet, as Patterson was getting at, are archives of Earth’s atmospheric history. At the poles, each year’s snowfall gets compressed into discrete layers of ice, in which little bubbles of air and dust are trapped. By drilling down and analyzing those layers, scientists can see how the chemical composition of our planet’s atmosphere has changed — is changing — over time. Today, this is common knowledge, the foundation of nearly all modern climatological research. But nobody had thought to look at ice cores this way before, not until Clair Patterson went looking for lead.


In 1964 and 1965, with funding from the National Science Foundation, Patterson drilled and collected ice cores from glaciers in both Greenland and Antarctica. Chemical analyses of these ice cores showed that global atmospheric lead levels had been stable for thousands of years, and had begun to increase, slightly, around 100 B.C. — a byproduct of Roman smelting and metalworking. It wasn’t until more recent times that lead levels had really begun to take off, however. Lead levels in our planet’s atmosphere exploded in the early-1920s — when leaded gasoline was released to market — and had been rising steadily and dangerously ever since. In 800 B.C., global atmospheric lead concentrations measured less than 0.001 micrograms per kilogram. By the mid-1960s, that figure had soared to more than 0.2 micrograms per kilogram — a 200-fold increase.
It was around then that Patterson began looking at lead levels in human beings. He compared the bones of 1,600-year-old Peruvian skeletons with those of contemporary Americans. (Lead accumulates in our bones and teeth throughout our lives.) Modern bones contained between 700 and 1,200 times more lead than ancient bones, Patterson found.
Patterson published these findings in the Archives of Environmental Health in 1965. The paper was titled “Contaminated and Natural Lead Environments of Man.” Drawing on data from ocean sediments, mountain snows, ice cores, and ancient versus modern human bones, Patterson demonstrated that industrial lead had polluted the entire planet. Atmospheric lead levels were particularly pronounced in the northern hemisphere (where the bulk of the world’s automobiles were located at the time) — about 1,000 times higher than natural levels. Human beings were being polluted, too. In 1965, the average American carried a blood lead level around 100 times greater than what should occur naturally — about 25 micrograms per deciliter compared to a natural baseline of 0.2 micrograms per deciliter. The average American, Patterson wrote, was being “subjected to severe chronic lead insult.” And this lead, Patterson demonstrated, wasn’t coming from volcanoes or forest fires or meteorites or other natural sources, as other scientists had surmised, but flowing from the tailpipes of hundreds of millions of automobiles.
An entire generation — the entire planet! — was being poisoned, Clair Patterson had discovered, and he knew exactly where the poison was coming from.
II. The Deadliest Man Who Ever Lived
Thomas Midgley Jr. was born on May 18, 1889, in Beaver Falls, Pennsylvania, into a family of inventors. His father, Thomas Midgley Sr., was an innovator in the field of automobile wheels and tires. His mother, Hattie Midgley, was the daughter of James Emerson, the man who invented the inserted-tooth saw. Maybe it was nature, or nurture, or both, but from an early age, the young Thomas Midgley displayed a knack for thinking outside the box. As a boy, he discovered that chewing the bark of slippery elm trees and applying the paste to baseballs gave them a more curved trajectory when thrown — a trick that big league pitchers would later adopt. In geometry class, he opted to solve problems by methods different from those in the textbook or demonstrated by his teacher, always looking for a better way. At Betts Academy, a prep school where Midgley prepared for college, a professor named H.M. Roberts introduced him to the tool he’d use to change the world: the periodic table of the elements.
After graduating from Cornell in 1911 with a degree in mechanical engineering, Midgley got a job designing cash registers at the National Cash Register Company in Dayton, Ohio. The work bored him, but taught him a few practical engineering skills. After about a year, Midgley went on to work as an engineer at his father’s wheel-and-tire manufacturing company. The family business went out of business shortly thereafter, however, and father and son had to find new employment. In 1916, Midgley Jr. was recruited by General Motors’ Dayton Engineering Laboratories Company, better known as Delco. He was twenty-seven years old. He would later say that the decision to join Delco was “the most important one of his whole life,” and that the years that followed were “like a story from the Arabian Nights.” Straight away, the company assigned him one of its most irksome problems: engine knock.

With the introduction of high-compression engines, the automotive industry faced a new challenge. Low-quality, unrefined gasoline would combust prematurely in engine cylinders, causing the engine to shake and shudder with sharp, unpleasant pinging sounds. Engine knock, as the problem was known, was annoying, damaging, and inefficient. If someone could solve it, there was enormous money to be made.
Midgley started testing compounds, searching for something that could eliminate engine knock. Early on — by accident, really — he discovered that adding iodine to fuel stopped the knocking. Iodine was an imperfect solution, however. It was corrosive and prohibitively expensive, adding more than a dollar to the cost of a gallon of gas.
Iodine was a dead end, but it was a turning point in Midgley’s research. It proved the problem could be solved with chemistry! Encouraged by the discovery, Midgley embarked on what he called a “scientific foxhunt.” He pinned a copy of the periodic table onto a pegboard in his laboratory and began mapping the anti-knock effectiveness of each element as it was tested. He tested hundreds — possibly thousands — of compounds over the next few years. He tested compounds of iodine and bromine, selenium and tellurium and tin. A pattern emerged: anti-knock effectiveness increased as he descended these groups in the periodic table and as he moved from right to left across a period.
By the fall of 1921, this systematic testing pointed to lead as the best candidate. A compound called tetraethyl tin had proven mildly effective at reducing engine knock earlier that year, and Midgley’s periodic table analysis predicted that lead — the heaviest stable element at the bottom of that column — would work even better. On December 9, 1921, Midgley had a small volume of tetraethyl lead delivered to the Delco lab. When he added it to gasoline and started the test engine, the knocking vanished instantly. Even diluted to a strength of one part tetraethyl lead to 1,300 parts gasoline, the compound had a remarkable ability to silence knocking engines. Midgley and his colleagues “danced a very un-scientific jig” and rushed to share the news with their boss, Charles Kettering. Midgley would later attribute the breakthrough to “luck and religion, as well as the application of science.”

The solution was miraculous.
It was also, everyone involved in its creation knew, highly toxic.
Lead’s dangers had been known for millennia by this time. Famously, the ancient Romans used lead pipes and added lead to wine; historical records are rife with descriptions of symptoms consistent with lead poisoning. “Water from clay pipes is much more wholesome than that which is conducted through lead pipes, because lead is found to be harmful for the reason that white lead [lead carbonate] is derived from it, and this is said to be hurtful to the human system,” the Roman architect Vitruvius wrote, two thousand years ago, during the reign of Emperor Augustus. “Hence, if what is produced from it is harmful, no doubt the thing itself is not wholesome.”
“This we can exemplify from plumbers,” Vitruvius continued, “since in them the natural colour of the body is replaced by a deep pallor. For when lead is smelted in casting, the fumes from it settle upon their members, and day after day burn out and take away all the virtues of the blood from their limbs. Hence, water ought by no means to be conducted in lead pipes, if we want to have it wholesome.”
In letters and essays from the mid- to late-1700s, Benjamin Franklin warned that lead caused “dry gripes” (severe abdominal pain) and paralysis. He also observed that workers exposed to lead fell ill more often than most; that lead poisoning seemed to be cumulative, worsening over time; and that even small, repeated doses seemed to be harmful.
In 1891, a British physician called Sir Thomas Oliver published Lead Poisoning in its Acute and Chronic Forms, which quickly became a standard medical reference. In the book, Oliver linked neurological damage, abdominal colics, paralysis, and long-term disability to lead exposure, and emphasized that the damage inflicted by the element was systemic, cumulative, and often irreversible. Oliver also documented cases of lead poisoning in folks working in silver mines and paint and ammunition manufacturing plants. These workers’ memories failed them, Oliver observed. Their cognition deteriorated. They grew anemic. Some suffered seizures. Some fell into comas. Some died.
General Motors knew they had a toxic product on their hands in tetraethyl lead, but the profits were too big to pass up. (They had also developed lead-free alternatives. During his “scientific foxhunt,” Midgley had developed an ethanol-based anti-knock agent which didn’t contain lead, but GM couldn’t patent it, and thus, couldn’t make any money from it. The company quietly shelved the finding.) On February 2, 1923, leaded gasoline was released to market — and into the environment. The following year, General Motors and Standard Oil (which later became Exxon) formally created the Ethyl Gasoline Corporation to manufacture and market the creation. The company carefully avoided any mention of lead in the product’s name or advertising. They simply called it “Ethyl.”

Midgley himself was intimately familiar with his invention’s toxicity. In 1923, after finding that lead formed corrosive deposits in engines, he added ethylene bromide to the fuel — a compound that caused the lead to be expelled completely through the exhaust and into the atmosphere. He inhaled vast quantities of the fumes, and, as a result, suffered a bout of lead poisoning so severe he had to take an extended vacation to recover. “I find that my lungs have been affected,” he wrote, “and that it is necessary to drop all work and get a large supply of fresh air.” He went to Florida. He wrote a paper on the health hazards of tetraethyl lead shortly thereafter. Yet, he never wavered in his conviction that Ethyl gasoline was safe — not publicly, anyway.
Midgley, the historian David Rosner has said, “had an enormous interest in self-delusion when it came to a product that he was so connected to. Of course, he had an ethical dilemma. Whether he fooled himself, lied, or was just oblivious to what future generations would have to deal with is beyond me.”
Midgley may have been the first person to be poisoned by his invention, but he certainly wasn’t the last. At GM’s prototype plant in Dayton, two workers packing bottles of tetraethyl lead on the assembly line dropped dead in April 1924. Dozens of other workers were sickened by lead poisoning that same month. The staff grew so demoralized they were, according to company records, “depressed to the point of considering giving up the whole tetraethyl lead program.” The line was shut down, but GM didn’t abandon the project. Instead, Charles Kettering, president of the General Motors Chemical Company at the time, blamed the workers themselves, claiming they “used to snap the stuff at each other, and throw it at each other, and they were saying that they were sissies. They did not realize what they were working with.” The company shifted tetraethyl lead production to other facilities, where more workers would die.
General Motors wasn’t the only company dealing in tetraethyl lead by this time. DuPont’s plant in Deepwater, New Jersey, along the Deleware River, began tetraethyl lead production in September 1923. Within a month, at least one worker had died of lead poisoning. When production ramped up in the summer of 1924 to meet soaring demand, three more workers died. Four more would perish in early 1925. Between 1923 and 1925, over 300 workers suffered lead poisoning at DuPont’s Deepwater plant. At least eight lost their lives. “Not surprisingly,” journalist Jamie Lincoln Kitman wrote in The Nation in 2000, “given DuPont’s stranglehold on all local media within its domain along the Delaware, the deaths went unreported.” (The workers, those who survived, called the Deepwater plant “The House of Butterflies” for the hallucinations they experienced there — another common symptom of lead poisoning.)
The disaster that finally broke through into public consciousness occurred in October 1924, at Standard Oil’s refinery in Bayway, New Jersey. Over a period of several days, workers started going mad. First came disorientation, then fits of violent rage, followed by bouts of hysterical laughter. The more delirious among them had to be wrestled into straitjackets. Five died. Thirty-five others suffered severe lead poisoning: tremors, hallucinations, violent palsies. Some would spend the rest of their lives in psychiatric institutions. One worker, Joseph G. Leslie, was quietly transferred to Graystone Psychiatric Hospital in New Jersey, where he would spend the next forty years. Only his wife and son knew the truth; the rest of the family believed he was dead.
New York City banned the sale of leaded gasoline shortly thereafter. So did Philadelphia, Pittsburgh, and the State of New Jersey. “Such mixtures of gasoline,” the New York City Board of Health stated, “containing lead or other deleterious substances, may be liable to prove detrimental and dangerous to the health and lives of the community, particularly when released as exhaust from motor vehicles.”
At Standard Oil’s first press conference about the Bayway disaster, a company spokesman claimed he knew nothing of the incident, then told reporters that “nothing ought to be said about this matter in the public interest.”

On October 30, 1924 — the same day New York City banned leaded gasoline — Thomas Midgley Jr. held a press conference of his own. Standing before a room of reporters, he poured tetraethyl lead over his hands, held a bottle of it under his nose, and inhaled its vapors for a full sixty seconds. “I’m not taking any chance whatever,” Midgley proclaimed. “Nor would I take any chance doing that every day.” This was the same Thomas Midgley Jr. who, just a year earlier, had been forced to flee to Florida to recover from severe lead poisoning — a condition from which, according to his own letters, he never fully recovered. (Considering this, historians have since questioned whether Midgley actually used tetraethyl lead in the demonstration; some think he might have used glycerine instead.)
Other industry reps echoed Midgley’s position: the deaths resulted from poor factory ventilation and worker carelessness, not the inherent toxicity of the product. One industry representative told reporters that the Bayway workers “probably went insane because they worked too hard.”
Right . . .
In May 1925, the U.S. Public Health Service convened a hearing. Frank Howard of Standard Oil asserted that leaded gasoline posed no threat to public health. Tetraethyl lead was diluted more than a thousand to one in gas! If you weren’t a factory worker exposed to huge amounts of the stuff, you had nothing to worry about. Public health experts pushed back. “There are thousands of things better than lead to put in gasoline,” a Harvard physician named Alice Hamilton said. She was correct, of course. Ethanol worked. So did other additives, some of which were patented by GM itself. (According to a 1957 memoir by Thomas Boyd, a chemist who helped Midgley develop leaded gasoline at Delco’s Dayton lab, Alice Hamilton confronted Charles Kettering in a hallway during one of the hearing recesses. “You are nothing but a murderer,” she told him. He laughed in her face.)
The Public Health Service, under heavy industry pressure, canceled a second day of hearings that would have explored these alternatives. On January 19, 1926, the agency issued a report finding “no good reason” to prohibit leaded gasoline, though it noted that “if the use of leaded gasoline becomes widespread, conditions may arise very different from those studied by us which would render its use more of a hazard than would appear to be the case from this investigation.” The report called for more research. No follow-up studies were funded by the Public Health Service. Brief bans instituted by places like New York City and Philadelphia and Pittsburgh and the State of New Jersey were lifted. Signs started popping up at gas stations: “ETHYL IS BACK.” Ethyl gasoline had prevailed. It became the standard fuel across America, and soon, the world.


For decades, research suggesting that lead was harmful was silenced or suppressed. In 1943, pediatric neurologist Randolph Byers and child psychologist Elizabeth Lord published a groundbreaking study showing that children who had suffered acute lead poisoning remained intellectually and behaviorally impaired into adulthood. Some of these children exhibited aggression so severe they were expelled from school, set fires to buildings, and assaulted classmates. The findings gained national attention in Time magazine, at which point the Lead Industries Association and its lawyers “descended upon” Byers in Boston and “threatened him with lawsuits,” says David Rosner. The industry then gave Byers a grant — or perhaps it was hush money — and for the next ten years, he published only two articles, neither of which had anything to do with lead.
By the mid-1960s, nearly 100 million motor vehicles were registered in the United States, and nearly all of them were running on Ethyl gas. Engine manufacturers designed new, more powerful engines specially for it — engines with higher compression ratios, greater horsepower, better fuel efficiency. The automotive industry boomed, pumping more than 200,000 tons of lead into the atmosphere each year.

As for Thomas Midgley Jr., well, he’d long since moved on to his other unfortunate invention.
In the early-1930s, GM’s refrigerator division, Frigidaire, had a problem on their hands. The gases used in early refrigerators — ammonia, methyl chloride, sulfur dioxide — were both highly toxic and highly flammable. These gases occasionally leaked, and killed entire families in their sleep. In 1929, one such leak at a hospital in Cleveland caused an explosion that killed more than a hundred people — doctors, nurses, patients. Midgley was tasked with finding a safe alternative.
Quickly, he hit upon dichlorodifluoromethane, which belongs to a class of chemical compounds called chlorofluorocarbons, or CFCs. The company marketed it as “Freon.” To demonstrate Freon’s safety, Midgley staged another publicity stunt where he inhaled a lungful of the gas and used it to blow out a candle, proving that it was neither toxic nor flammable. Freon became ubiquitous in refrigerators, air conditioners, and aerosol cans. Midgley was lauded as a hero. He was awarded the Priestley Medal in 1941 and named president of the American Chemical Society shortly thereafter. If the United States had a Royal Family, he probably would have been knighted.
What Midgley didn’t know — what no one knew — was that dichlorodifluoromethane and other CFCs rise into the stratosphere and destroy the ozone layer that shields all life on Earth from ultraviolet radiation. CFCs are also potent greenhouse gases, trapping heat far more effectively than carbon dioxide. It would be decades before scientists understood the damage.
Thomas Midgley Jr. would be dead by then. He contracted polio in 1940. The virus left him paralyzed. Ever the engineer, he designed an apparatus of ropes and pulleys to lift himself out of bed without assistance. On November 2, 1944, he became entangled in the device, and strangled to death. The coroner ruled it a suicide. He was fifty-five years old.

Environmental historian John McNeill would later write that Midgley “had more impact on the atmosphere than any other single organism in Earth’s history.” In his book, A Short History of Nearly Everything, Bill Bryson wrote that Midgley possessed “an instinct for the regrettable that was almost uncanny.” In May 2010, Time magazine listed both leaded gasoline and CFCs among the fifty worst inventions of all time.
III. Unleaded
Perhaps this story should have been called “A Tale of Three Scientists,” because there’s another scientist — a toxicologist, specifically — who figures prominently within it.
His name was Robert Kehoe, and he was a professor of occupational medicine at the University of Cincinnati. In 1924, he was hired by Charles Kettering of General Motors to study the health effects of tetraethyl lead. In 1925, he was appointed chief medical advisor to the Ethyl Gasoline Corporation. In 1930, with funding from General Motors, Ethyl, and DuPont, he built and became director of the Kettering Laboratory of Applied Physiology at the University of Cincinnati — one of the nation’s first university-based laboratories devoted to studying industrial toxicological problems. For nearly half a century, almost all research support concerning the dangers of tetraethyl lead — and, in turn, leaded gasoline — came from industry and was funneled to Kehoe. He developed the “Kehoe Rule,” or “Kehoe Paradigm” — the idea that lead levels below 80 micrograms per deciliter of blood were perfectly safe — and argued that lead levels in modern humans were perfectly natural.

In the decades between Thomas Midgley Jr.’s invention of leaded gasoline and Clair Patterson’s discovery of global lead pollution, Kehoe’s word on all things lead was gospel. He was the leading figure in the field, and there wasn’t a close second. He was awarded the presidency of the American Academy of Occupational Medicine and directorship of the Industrial Medical Association, among other accolades too numerous to list here. The Archives of Environmental Health dedicated an entire issue in honor of his work.
That’s the same journal, you may recall, in which Clair Patterson published his paper, “Contaminated and Natural Lead Environments of Man,” in 1965.
That must have been awkward.
“The issue which he has raised, in this article and by word of mouth elsewhere, cannot be ‘swept under the rug,’” Kehoe stated in response to Patterson’s paper. “It must be faced and demolished, and therefore, I welcome its ‘public appearance.’” And he continued: “The inferences as to the natural human body burdens of lead, are, I think, remarkably naïve. It is an example of how wrong one can be in his biological postulates and conclusions, when he steps into this field, of which he is so woefully ignorant and so lacking in any concepts of the depths of his ignorance, that he is not even cautious in drawing sweeping conclusions.” Kehoe’s message to Patterson was clear: Stick to the rocks, nerd. Stay in your lane. Leave the toxicology to the toxicologists.
Others piled on. “Is Patterson trying to be a second Rachel Carson?” Herbert Stockinger, a colleague of Kehoe’s, sneered. He went on to refer to Patterson’s research as “science fiction.”
Patterson ignored them, as best he could. He started writing letters to lawmakers, trying to spread the word about the environmental crisis he’d uncovered. California Governor Pat Brown politely declined Patterson’s concerns. But Senator Edmund Muskie of Maine, chairman of the Subcommittee on Air and Water Pollution, paid attention. On October 7, 1965, Patterson wrote to Muskie offering to testify about his research and its implications. Muskie invited him to a hearing on June 15, 1966.

The 1966 hearing brought together leading scientists from both sides, including Patterson and Kehoe. It would become one of the most consequential confrontations in the history of environmental and public health.
During the hearing, Kehoe was hesitant to go into too much detail about his research. It was far too complicated to be comprehended by lawyers and policymakers — laymen. “I’m afraid we would be here the rest of the week if I were to undertake to do this,” he said. But Muskie pressed him. Kehoe insisted that atmospheric lead levels hadn’t increased as a result of leaded gasoline; that current levels of lead in the environment and in human bodies were natural, and human beings had “adapted” to them; that factory workers were the ones who really had to worry about lead exposure, and they already received adequate protection. When Muskie asked, “Does medical opinion agree that there are no harmful effects and results from lead ingestion below the level of lead poisoning?” Kehoe replied, “I don’t think that many people would be as certain as I am at this point.”
“But you are certain?”
“It so happens,” Kehoe said, “that I have more experience in this field than anybody else alive.”
What Kehoe really stressed, over and over again, was that the science wasn’t settled. Was it possible that humans had increased atmospheric lead concentrations, a little bit? Sure, but it hadn’t been demonstrated by anyone other than that kook Clair Patterson. More research was needed. Was it possible that modern atmospheric lead levels could harm people? “It is entirely possible to postulate that somewhere, sometime, within the universe of the population, you will find individuals who are so especially susceptible to lead that, under the conditions that exist in the rest of the population, that these people will be singled out and poisoned,” Kehoe explained. Was it possible that blood lead levels below 80 micrograms per deciliter were harmful? Maybe, but Robert Kehoe — the Robert Kehoe — had “never seen one case of lead poisoning” below this level.
In effect, Kehoe parroted what lead industry reps had been saying since the 1920s: large doses of tetraethyl lead were potentially harmful, if mishandled, but leaded gasoline was perfectly safe.
Then came Patterson’s testimony. He came out swinging. He cited his ocean sediment data, his ocean water data, his ice core data showing that atmospheric lead levels had surged since human beings started burning leaded gasoline. “Because of this extensive pollution,” he said, “typical lead levels observed in our environment today may be grossly higher than natural lead levels. It is important therefore to carefully distinguish between the typical levels of lead in our environment and those natural levels which refer to the levels of lead in body and environment which prevailed during the creation and the evolution of our physiological responses to lead.”
This may have been Patterson’s most important point, the most important point: that there was a difference between “natural” and “typical” levels of lead. It was a distinction that Clair Patterson had spent the last decade studying. It was news to industry-funded researchers like Robert Kehoe and institutions like the U.S. Public Health Service, however. Scientists and laymen alike needed to update their lexicon. The lead in modern bones and bloodstreams wasn’t natural, but it was typical, only because, in the words of Clair Patterson, “Earth’s entire biosphere” had been “heavily contaminated with industrial lead.”
It was a simple — but crucial — distinction, Muskie agreed. “Now, why has it [this distinction] not been attempted by these organizations or by others than yourself in studying this problem?” he asked Patterson. “It seems such a logical approach to a lawyer.”
“Not if your purpose is to sell lead,” Clair Patterson said.
That raised a few eyebrows.
“Well,” Muskie said, “I don’t think it is the purpose of the Public Health Service to sell lead.”
“That is why it is difficult to understand why the Public Health Service cooperated with the lead industry in issuing this report which fails to make this distinction.” Patterson is referring here to a 1965 study carried out by the U.S. Public Health Service — in collaboration with the American Petroleum Institute, the Automobile Manufacturers Association, Ethyl Corporation, and the Kettering Laboratory of Applied Physiology — which concluded that modern atmospheric lead levels were “normal” and posed no threat to human health. Clair Patterson had taken the gloves off.

“Have you discussed this question with anyone in the Public Health Service?” Muskie asked.
“Yes.”
“What is their reaction?”
Patterson shrugged. “I have both friends and antagonists in the Public Health Service,” he said.
Patterson supplied written testimony as well. He wrote:
The latest view of existing lead states in this country, officially sanctioned by the United States Department of Health, Education and Welfare, is that they “are well within the presently accepted range of lead levels for humans and are not significant in terms of a threat of the occurrence of lead intoxication.” This view has prevailed in the state and the Federal Public Health Services for decades. It is based upon a threshold for damage concept [the “Kehoe Rule”] which has been applied to industrial workers, and which involves the axiom that a worker must be either perfectly healthy or classically intoxicated with lead but cannot be neither. This is a seriously unfortunate situation.
He went on to explain that the average American blood lead level of 25 micrograms per deciliter sat about a third of the way between a natural level of 0.2 micrograms per deciliter and Kehoe’s toxicity threshold of 80 micrograms per deciliter. Americans weren’t just exposed to a little bit of lead here and there, Patterson made clear — they were carrying dangerous levels in their bloodstreams. They were, in Patterson’s words, “subjected to severe chronic lead insult.” They were being poisoned, and the Public Health Service was doing nothing about it. The Public Health Service was letting the industry police itself. The Public Health Service was collaborating with the lead industry, toeing the industry line.
“It is not just a mistake for public health agencies to cooperate and collaborate with industries in investigating and deciding whether public health is endangered,” Patterson concluded, “it is a direct abrogation and violation of the duties and responsibilities of those public health organizations . . . Whether the best interests of the public have been served by having public health agencies work jointly with representatives of lead alkyl industries in evaluating the hazards of lead alkyls to public health is a question that should be asked and answered.”
Clair Patterson was pretty punk rock, but he paid a price for speaking truth to power. Following the 1966 hearing and his criticisms of America’s public health agencies, research organizations — including, unsurprisingly, the U.S. Public Health Service — refused him contracts. Robert Kehoe and his colleagues did their best to tarnish his reputation. In 1971, the Environmental Protection Agency contracted with the National Academy of Sciences to assemble an expert panel on atmospheric lead pollution. Patterson wasn’t invited, but Kehoe was. The panel’s report, “Airborne Lead in Perspective,” ignored Patterson’s research entirely. He vented in a letter to Harrison Brown: “Lawyers are not scientists and neither are government bureaucrats — and when the bureaucrats are elected by people, the majority of whom believe in astrology and do not believe in evolution, then this sort of thing can be expected.”
Patterson’s words got through to his fellow scientists, however. Almost immediately, other researchers started replicating his findings. (One team looking at atmospheric lead levels during the Roman period carried out ice core analyses of their own, then called him up and said, “Pat, we’ve got your curve! It’s the same damn curve!”) Some began to build upon them. In October 1967, a pair of environmental epidemiologists from the California State Department of Public Health published a study in Science finding a dose-response relationship between atmospheric lead levels and human blood lead levels. The team examined general and occupational populations. The pattern held across epidemiological and experimental data. The paper concluded that “long-term increases in atmospheric lead will result in predictably higher blood lead levels in the exposed populations.” What Clair Patterson had evinced through ice cores and mummy bones now had a quantifiable mechanism. (Meanwhile, a pediatric psychiatrist named Herbert Needleman was following in the footsteps of Randolph Byers and Elizabeth Lord, conducting research linking elevated blood lead levels in children with reduced IQ and other developmental issues. In 1979, Needleman published a study in the New England Journal of Medicine showing that these adverse effects can occur at blood lead levels below Kehoe’s poisoning threshold of 80 micrograms per deciliter.)
Patterson’s words got through to Edmund Muskie, too. In 1970, Senator Muskie shepherded the Clean Air Act through the Senate with unanimous support, giving the newly formed Environmental Protection Agency authority to regulate fuel additives. The EPA moved quickly. In January 1973, the agency announced that gas stations would be required to offer unleaded gasoline by mid-1974. Later that year, the agency issued regulations limiting the lead content of all grades of gasoline to 1.7 grams per gallon by July 1975 — a first step in a gradual phasedown toward the long-term target of 0.1 grams per gallon. The industry sued, argued that the science wasn’t settled. In the landmark 1976 case Ethyl Corp. v. EPA, Judge J. Skelly Wright ruled that “awaiting certainty will often allow only for reactive, not preventive, regulation.” He allowed the phasedown to proceed.



Quickly, lead began to vanish from American air. In the late-1960s and early-1970s, more than 200,000 tons of lead went into gasoline — and into the atmosphere — annually. By 1988, that figure had fallen below 4,000 tons. Between 1976 and 1980, as the lead content of gasoline was cut in half, American blood lead levels fell 37 percent. Things were moving in the right direction.
In 1978, the Consumer Product Safety Commission banned lead-based paint for residential use (another common route of lead exposure). That same year, Clair Patterson was finally admitted to a National Research Council panel on atmospheric lead pollution. While the majority recommended more study before action, Patterson wrote a seventy-eight page minority report arguing for the immediate removal of lead from gasoline, food containers, paint, and water pipes. He’d spent his career being told he was too eccentric, too intense, too extreme. He refused to soften his message.
Children’s blood lead levels fell nearly 80 percent between the late-1970s and early-1990s. In 1985, the EPA mandated a more than 90 percent reduction of lead content in gasoline. By 1986, lead levels in gas had dropped to 0.1 grams per gallon. Lead solder was removed from food containers. Lead pipes began to be replaced in water distribution systems. Finally, at last, the Clean Air Act Amendments of 1990 required all leaded gasoline to be removed from service stations by the end of 1995. Clair Patterson’s fifty year war on leaded gas was nearing its end.
Patterson was awarded the Tyler Prize for Environmental Achievement that same year, in April 1995. The award recognized his research demonstrating the pervasive health risks of global lead pollution, his influence on regulatory change to reduce lead pollution, and his prolonged efforts on behalf of the environment. He sat down with Shirley Cohen shortly thereafter, and told her about his life. Cohen asked him if he had any pride in having gotten the award, in having his work vindicated after all these years.

“I don’t have any pride, I’m sorry to say,” Patterson told her. “I have zero pride in any award. All I feel is obligation, obligation, and obligation. I’m sorry, but that’s my personality. I feel obligated and obliged.”
“Instead of the word ‘pride,’ can I use the word ‘pleasure,’ gratification?” Cohen asked.
“No, not gratification,” Patterson said. “No, it is an awareness of the worthiness of a communal spirit of science. It’s not a personal thing at all.”
“Can Laurie be proud of you?”
“Oh, yes. But it’s very difficult for her to get along. I certainly wouldn’t marry myself if I were her. Bless her heart.”
They laughed.
“Would you have [done] anything different if you could do it again?” Cohen asked, bringing the interview to a close.
“Well,” Patterson said, “there’s a Nobel laureate poet [novelist and philosopher], Camus. He wrote an essay called ‘The Myth of Sisyphus.’ It dealt with suicide — about being alive or dead. I’m a manic depressive, of course, because I’m very, very depressed overall, all the time. But being alive physically is only a manifestation of being alive emotionally for 100,000 years. You see, we don’t die really. Physically we do, perhaps. But we’re part of a whole. We’re a unit; we’re Homo sapiens sapiens. We are brain containers. This thing generates thinking and emotions. Here’s the important thing, up here. Now, right now, the way things are right now, the emotions within an individual brain can’t be communicated. You don’t know the emotion of the artist who composed that music. We do not know . . .”
Clair Cameron Patterson died a few months later, on December 5, 1995, at his home in Sea Ranch, California. It wasn’t suicide that killed him, but an asthma attack. He was seventy-three years old.
Three weeks later, on January 1, 1996, the United States banned the sale of leaded gasoline for on-road vehicles.
IV. Fallout
It wasn’t until decades after Clair Patterson’s death — quite recently, in fact — that scientists began to understand just how severe the environmental crisis he uncovered truly was. The damage, we now know, was far worse than even Patterson had suspected.
In 2022, researchers at Duke and Florida State University published a study in the Proceedings of the National Academy of Sciences attempting to quantify the cognitive damage leaded gasoline inflicted on Americans. Drawing on blood-lead data, gasoline consumption records, and population statistics, they estimated that, as of 2015, lead pollution had stolen 824 million IQ points from more than 170 million Americans — about half of the U.S. population. The average loss was 2.6 IQ points per person, but damage varied by birth year. Those born in the mid-to-late 1960s, at leaded gasoline’s peak, lost an average of about six IQ points. Some lost more than seven points. These aren’t abstract numbers. You, dear reader, could very well be one of these statistics. So could your parents, or your parents’ parents.
“I frankly was shocked,” Michael McFarland, the study’s principal author, said in a press release announcing the findings. “And when I look at the numbers, I’m still shocked even though I’m prepared for it.”
IQ loss is just one of leaded gasoline’s lingering side effects, however. One of many. Lead’s damage to human health is disturbingly comprehensive. The metal affects nearly every organ system in the body; there is no “safe” level of lead in human blood. It’s probably carcinogenic to humans, according to the International Agency for Research on Cancer. It causes chronic kidney disease. It causes cardiovascular disease: hypertension, heart attacks, strokes, peripheral artery disease. It damages the hematological system, interfering with the production of hemoglobin and causing anemia. The list goes on.
In the brain, lead’s effects are structural and permanent. Brain imaging studies have revealed that adults exposed to lead as children show reduced gray matter volume — particularly in regions responsible for memory and executive function — along with compromised white matter integrity. Simply put, their brains look physically older than they should. A 2020 study of New Zealanders found that those with high childhood lead exposure had measurably “older” brains at age forty-five, as determined by MRI scans.
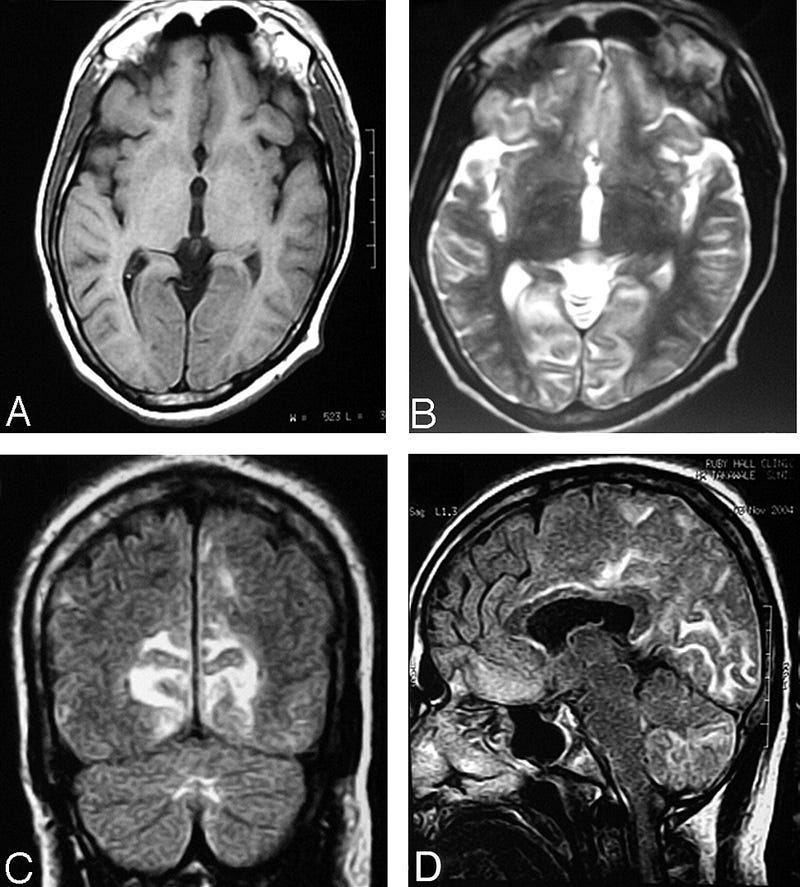
Lead exposure in childhood has been linked to a cascade of mental health problems that last into adulthood. These include major depression, panic disorder, generalized anxiety disorder, ADHD, and behavioral problems including aggression and difficulty with impulse control, just to name a few. Our personalities, even, are altered by lead: adults exposed to lead as children tend to score lower on conscientiousness and emotional stability, for example.
Lead exposure in childhood has been linked with violent crime as well, with research suggesting a lag of approximately twenty years between exposure and elevated crime rates. This timeline places peak lead exposure in the 1960s and 70s alongside the crime waves of the 1980s and 90s. Lead didn’t just make us less intelligent; it may have made us more violent, too.
Most insidious, perhaps, are lead’s emerging links with neurodegenerative diseases, including dementia. Children born in the 1960s and 70s — those with the highest lead exposure levels in human history — are now entering the age when dementia symptoms typically emerge. Lead stored in bones may be remobilized in old age, particularly during menopause or with osteoporosis, released into the bloodstream to attack an already vulnerable aging brain. Research presented at the 2025 Alzheimer’s Association conference found that Americans who lived in areas with high atmospheric lead levels between 1960 and 1974 were 20 percent more likely to experience memory problems later in life than those who lived in areas with low atmospheric lead levels.
“Millions of us are walking around with a history of lead exposure,” says Aaron Reuben, co-author of that 2022 IQ study mentioned a few paragraphs back. “It’s not like you got into a car accident and had a rotator cuff tear that heals and then you’re fine. It appears to be an insult carried in the body in different ways that we’re still trying to understand but that can have implications for life.”
And Americans weren’t — aren’t — the only ones affected by lead pollution, of course. A 2023 study analyzing 2019 data estimated that, in that year alone, lead exposure from all sources caused 765 million lost IQ points in children under five and 5.5 million cardiovascular deaths in adults worldwide. The economic cost was estimated at six trillion dollars.
Removing lead from gasoline is now recognized as one of humanity’s greatest environmental and public health achievements. But the legacy persists. “Unlike such carcinogens and killers as pesticides, most chemicals, waste oils and even radioactive materials, lead does not break down over time,” Jamie Lincoln Kitman wrote in The Nation. “It does not vaporize, and it never disappears.” Thus, the seven million tons of lead that we’ve pumped into our planet’s atmosphere since the 1920s remains with us today, contaminating air, water, and soil and bioaccumulating in the tissues of plants, animals, and people. A report from Britain’s Royal Commission on Environmental Pollution concluded that humans “have dispersed lead so widely that it is doubtful whether any part of the earth’s surface or any form of life remains uncontaminated by anthropogenic lead.” The report was issued in 1983, but that statement remains true today.
We — all of us — continue to bear the health effects of Thomas Midgley Jr.’s toxic creation. We’re still tallying the costs. And we’re still burning leaded gasoline. A little bit of it, at least. Aviation fuel used by small private planes still contains lead, making it the largest remaining source of lead pollution in the United States.
Most developed nations banned or completed phaseouts of leaded gasoline for on-road vehicles in the 1990s. (Some were ahead of the curve. Japan, for example, banned leaded gasoline in 1986.) But in developing nations, industry corruption delayed action. Innospec, which by the early aughts was the world’s only manufacturer of tetraethyl lead, paid bribes to keep the product on the market, even as the science of lead’s harm was settled and safer alternatives existed. Between 2000 and 2008, the company paid approximately $2.9 million in bribes to Indonesian government officials to secure tetraethyl lead sales and delay the country’s phase-out. In Iraq, Innospec paid over $10 million in kickbacks to officials, including $150,000 specifically to ensure that a competing lead-free product was not approved for use in Iraqi refineries. Internal emails revealed the scheme’s cynicism: “We are sharing most of our profits with Iraqi officials. Otherwise, our business will stop and we will lose the market. We have to change our strategy and do more compensation to get the rewards,” one agent wrote. The company’s business director replied saying, “the fewer words the better!” In 2010, Innospec pleaded guilty to violating the Foreign Corrupt Practices Act, defrauding the United Nations, and violating the U.S. embargo against Cuba. The company paid approximately $40 million in fines. Four executives were later convicted in the UK’s first contested overseas corruption trial.
In 2002, at the World Summit on Sustainable Development in Johannesburg, South Africa, the United Nations launched the Partnership for Clean Fuels and Vehicles — an unprecedented public-private alliance that brought together environmentalists, scientists, government officials, and oil industry executives to eliminate leaded gasoline worldwide. All of sub-Saharan Africa went unleaded by January 1, 2006. Remaining holdouts — clustered in North Africa, the Middle East, Central Asia, and Eastern Asia — would take another fifteen years to persuade. Some faced challenges of war and instability; others, like Indonesia and Cuba and Iraq, were targets of Innospec’s bribery schemes. Eventually, Algeria became the world’s last nation running on leaded gas. The country exhausted its supply in July 2021. On August 30, 2021 — nearly a century after Midgley’s breakthrough — the UN declared the end of leaded gasoline for automobiles worldwide. The global phase-out is estimated to prevent 1.2 million premature deaths per year and save $2.4 trillion annually in healthcare costs and other expenses.
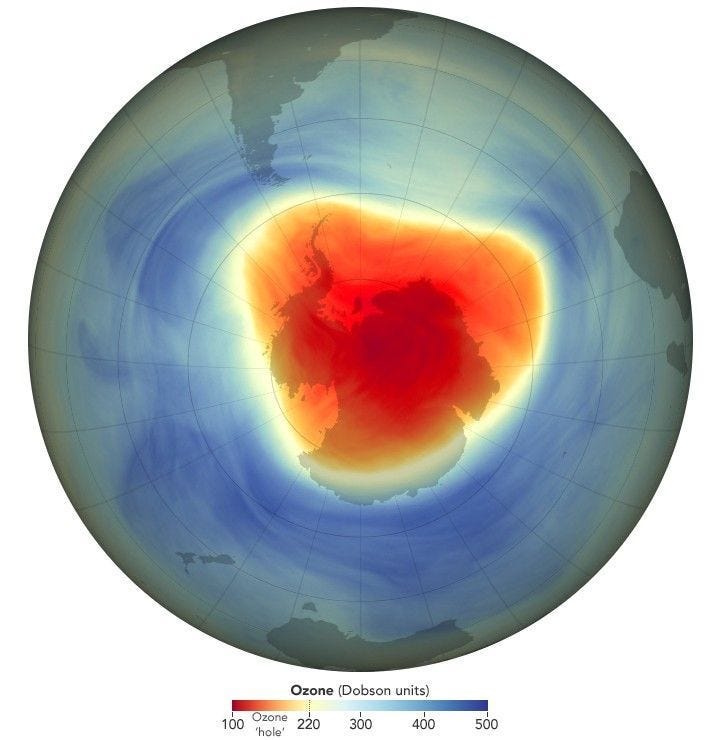
The ozone layer, damaged by Midgley’s other regrettable invention, CFCs — which were outlawed by the Montreal Protocol in 1987 — isn’t expected to make a full recovery until the 2060s.
V. The Communal Spirit of Science, and the Diseased Homo Sapiens Sapiens Mind
It’s impossible to quantify the full extent of the havoc that Thomas Midgley Jr.’s creations wreaked — continue to wreak — on Earth and its inhabitants.
Likewise, it’s impossible to quantify the number of lives that Clair Patterson’s research — and his courage to speak the truth — saved. In celebration of Patterson’s 1995 Tyler Prize for Environmental Achievement, marine chemist Edward Goldberg wondered aloud how many “additional years of life to world citizenry” could be attributed to him. The number is incalculable. There are too many variables! But it’s no exaggeration to say that each and every one of us has lived a longer, healthier life than we otherwise would have, if it wasn’t for Clair Patterson. “The blood lead levels of today’s children,” Herbert Needleman has written of Patterson, “are a testimony to his brilliance and integrity.”
The story of Clair Patterson and Thomas Midgley Jr. is many things. It’s a story about corporate and government corruption and the pursuit — and prevailing — of truth. It’s a story about one of humanity’s greatest environmental disasters, and one of our greatest triumphs. It’s a story about the “communal spirit of science,” in Clair Patterson’s words — the spirit that we’re all in this together, on this pale blue dot of ours, that we are all part of a whole — and the difference that one human being can make, for better or worse.

The story of Clair Patterson and Thomas Midgley Jr. is a paradox. Both men were scientists. Both men loved science, lived and breathed it. One man used science to poison the planet. The other used science to save it. Well, what are we supposed to make of that? What conclusions are we to draw about science and its role in our society? Is science, on the whole, a force for good, or a force for evil?
Toward the end of his life, Clair Patterson thought deeply about these questions. “Beginning in the early 1980s,” Patterson’s friend and colleague George Tilton wrote,
Patterson’s interests began to turn toward what I call the third stage of his intellectual career. It involved an introspective, philosophical evaluation of the place of man (H. s. sapiens, as he often stated it) in society. He distinguished between what he termed the engineering versus the scientific modes of thinking . . . He sees the scientific mind as the inquiring mind that seeks to uncover the world’s secrets, while the engineering mind seeks to control the natural world. This undoubtedly grew out of his experience as a scientist in discovering the age of the Earth, while the engineering mind would be equated with the technology that utilized the large amounts of lead that had polluted the environment. Thus he says, “Most persons cannot see the ills of a culture constructed by 10,000 years of perverted utilitarian rationalizations because they perceive only its material technological forms through the eyes of a diseased Homo sapiens sapiens mind.” At the end he was working on a book to express his ideas on those and other matters . . . We will never know what it might have contained, but we can guess that it would have been a stimulating, unique, and undoubtedly controversial treatment.
Like I said, Clair Patterson was pretty punk rock.
And I think he was onto something with that “scientific” versus “engineering” modes of thinking thing. In his interview with Cohen, Patterson used the words “utilitarian” and “nonutilitarian,” but the idea is the same. A scientist seeks to understand nature; an engineer seeks to control it. Scientists — true scientists — are nonutilitarian in their thinking; engineers are utilitarian.
“A scientist considers his data objectively because he has no material purpose,” Patterson stated in his 1966 testimony. “To him, science is an activity of the mind — the formulation of ideas concerning observations, the seeking of new observations, the revision of ideas — it is an inherently dynamic process of formulation and revision.”
“The utilization of observations for material purposes is not science,” Patterson went on. “It is engineering; it is the defense and promotion of industrial activity . . . This utilization is not done objectively. It is done subjectively.”
It’s a distinction that seems to have defined the difference between Clair Patterson and Thomas Midgley Jr.
Clair Patterson was a scientist — a true scientist — through and through. He devoted his entire life to understanding the world around him. The boy who asked his mother why a drop of water was round went on to determine the age of the Earth. Clair Patterson’s pursuit of understanding for understanding’s sake gave us superclean laboratories that revolutionized the way science of all varieties is conducted, and ice core analyses that laid the groundwork for modern climatological research. Clair Patterson helped us, as a species, better understand ourselves and our place in the cosmos, and gave us tools and techniques to continue refining our understanding of the world around us — the world we are part of — and what we are doing to it.
Thomas Midgley Jr. was, in Patterson’s framing, no scientist at all. He was an engineer, and a brilliant one at that. He was ingenious at taking science and utilizing it to solve specific, human-centric problems. And he solved real problems! Engine knock bedeviled early automobiles, and toxic refrigerator leaks were blowing up hospitals and killing people in their sleep. His solutions worked, too, at least in a narrow sense. Cars ran more smoothly. Refrigerators ran more safely. His inventions made human lives easier, more comfortable, and more convenient — at the expense, it turned out, of human health, and the health of the planet itself.
It’s easy to portray Thomas Midgley Jr. as a villain, but the truth, it appears, is more complicated than that. By all accounts, it seems that Midgley had the best of intentions, that he truly believed he’d done humanity a favor by inventing CFCs and leaded gasoline. A month before he died, in a presidential address to the American Chemical Society, Midgley recited a poem that he’d written. The poem reads like a kind of self-written eulogy (or, perhaps, considering the coroner’s ruling on Midgley’s cause of death, a suicide note). It reads:
When I feel old age approaching,
And it isn’t any sport,
And my nerves are growing rotten,
And my breath is growing short,
And my eyes are growing dimmer,
And my hair is turning white,
And I lack the old ambitions
When I wander out at night,
Though many men my senior
May remain when I’m gone,
I have no regrets to offer
Just because I’m passing on.
Let this epitaph be graven
On my tomb in simple style:
This one did a lot of living
In a mighty little while.
If Midgley was aware of the damage his inventions would inflict on our species and our planet, he left no trace of it. The man died with no regrets.
How, after all, could he have been?
How could he have known what we know today?
Certainly, Midgley knew that large doses of lead were harmful; he’d poisoned himself with a large dose of lead, after all. But it seems that he, like just about everyone else on planet Earth at the time, believed that low but chronic doses of lead stemming from leaded gasoline were nothing to worry about. Perhaps he was fooled by experts like Robert Kehoe. Or perhaps, as David Rosner suggests, he fooled himself.
Thomas Midgley Jr. wasn’t evil. I don’t think he was, at least. I think he was ignorant, short-sighted, and, in a sense, small-minded. I think he fooled himself. But, at the end of the day, when you boil it all down, it seems to me that he was guilty of nothing more than failing to see the bigger picture. Of failing to keep asking questions. Of failing to foresee the consequences of his actions, before he acted. Of failing to see that we, each and every one of us, are part of a whole; that when we harm the planet, we inevitably harm ourselves; that the health of humanity is inextricably intertwined with the health of the planet on which we live.
And aren’t we all? Aren’t we all, collectively, as a species, guilty of the same mistake — or similar mistakes, at least? Aren’t we all, collectively, as a species, burning fossil fuels and destroying habitat and driving other species extinct and befouling the planet with PFAS and microplastics and God-only-knows how many other pollutants, and only just beginning to understand how grave these mistakes may be, for us and the planet itself? And when confronted with the evidence of our mistakes and their consequences, don’t we persist in making them? And don’t we find countless ways to explain it all away, to justify what we are doing to ourselves and the planet . . . Aren’t we all, collectively, as a species, fooling ourselves?
This, I think, is the disease of the Homo sapiens sapiens mind that Clair Patterson alluded to: short-sightedness, self-centeredness — human-centeredness — coupled with ignorance, willful or otherwise, of our interconnectedness with everything else. That, and our propensity to believe our own bullshit.
Thomas Midgley Jr. was human, is what I’m trying to say. That may be the most unsettling thing about him. But we can’t blame him for his humaness, any more than we can blame ourselves for ours. It wasn’t until fairly recently, after all, in the grand scheme of things, that we humans began to realize the power we wield, for good or for ill. The notion that we, a band of apes gone loco, can decide the fate of the planet — that we are deciding the fate of the planet — is a relatively new idea. The notion that we can change the world — that we are changing the world, for better or worse — takes some getting used to. The situation in which we find ourselves is completely absurd.
Odd as it might sound, I think we owe Thomas Midgley Jr. a debt of gratitude, for he taught us, through his mistakes, just how destructive we humans can be, intentionally or unintentionally, consciously or unconsciously. He was the yin to Clair Patterson’s yang, and together, they raised the consciousness of the human race.
We learned something about ourselves, from these two scientists’ lives and work. We learned a lot about ourselves. Not least, we learned that, when confronted with the evidence of the harm we’re doing to ourselves and the planet, eventually, we are willing to change our ways; that we are willing to act for the good of the whole — the whole that we, Homo sapiens sapiens, are part.
Quentin Septer is a freelance science and environmental journalist based in Bogotá, Colombia. His writing has appeared in WIRED, Science, and Scientific American, among other publications.



Another reason to love my bicycle and imagine the benefits. That illusion still feels good!
The remaining IQ points I have (born in 1962) are pinging with stimulation over your extraordinary study of two men of science--and the roads they took. I appreciate your note about how the miracles (or the catastrophes) begin at the mandate of finding something useful to do with a major discovery. Useful to whom? And for how long? And for what price?